Covid-19 vaccine third dose approved for immunocompromised people
Spain's public health committee accepts it in very specific cases, but says no evidence that advises it for general population
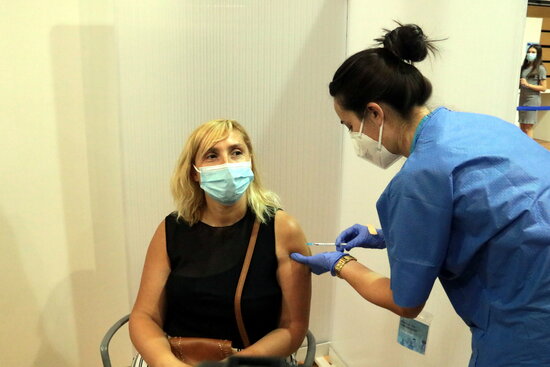
The Covid-19 vaccine third dose has been approved in Spain for immunocompromised people.
On Tuesday evening, the public health committee - formed by Spain's health ministry and regional health ministries - accepted it in very specific cases, such as solid organ transplant recipients, those having received hematopoietic stem cells due to diseases like leukaemia and people being treated with anti-CD20 medicines, common to fight diseases like lymphomas.
The third jab will have to be administered 28 days after the second, but in the latter case, the patient will have to wait six months after they finished their treatment.
MRNA vaccines will have to be used for all three groups, such as Pfizer or Moderna.
Officials are still looking into giving the booster dose to other immunocompromised people, such as certain cancer patients.
Yet, authorities believe there is no evidence that advises the third jab for general population.
As of September 7, 2021, 5,752,140 residents have been given the first dose of the vaccine, 72.7% of the total population. Out of those, 4,846,719 have also been administered a second dose (61.4% of the total population). 5,478,324 residents are considered to be fully immunized (69.3%). Under 65s who have already had the virus are only required to have the first dose, and others have received a single-dose jab. Therefore, the % of two doses administered and % fully immunized do not match.